|
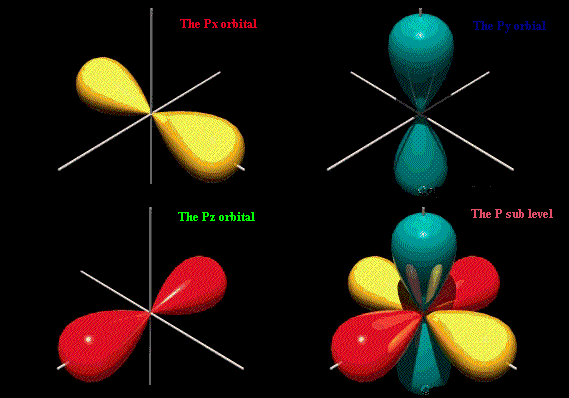
There is 25% s-character and 75% p-character in each sp 3 hybrid orbital.The four sp 3 hybrid orbitals so formed are directed towards the four corners of the tetrahedron. Sp 3 hybridisation: This type of hybridisation can be explained by taking the example of CH 4molecule in which there is mixing of one s-orbital and three p-orbitals of the valence shell to form four sp 3 hybrid orbital of equivalent energies and shape. Types of Hybridisation - sp 3 hybridisation
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
